Seznamy Hydrogen Atom Mass Čerstvý
Seznamy Hydrogen Atom Mass Čerstvý. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h.
Nejchladnější 1
The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. For hydrogen we have z = 1. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is the most abundant of all elements in the universe.It is named after niels bohr, due to its role in the bohr model of an atom.
The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. For hydrogen we have z = 1. Hydrogen is the most abundant of all elements in the universe. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; Hydrogen has isotopes whose natural quantities cannot be ignored. Its value is 5.291 772 109 03(80) × 10−11 m.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. We now have the tools to study the hydrogen atom, which has a central potential given by. The difference between the neutron number and the atomic number is known as the neutron excess: Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. Diagram of a simple hydrogen atom. Hydrogen is the most abundant of all elements in the universe. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; For hydrogen we have z = 1. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Hence one proton (hydrogen assuming negligible. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. For hydrogen we have z = 1. We now have the tools to study the hydrogen atom, which has a central potential given by.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Dec 08, 2020 · protons and neutrons in hydrogen.. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.

Hydrogen is the most abundant of all elements in the universe... Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. It is named after niels bohr, due to its role in the bohr model of an atom. Dec 08, 2020 · protons and neutrons in hydrogen. Hydrogen has isotopes whose natural quantities cannot be ignored. Diagram of a simple hydrogen atom.. Its value is 5.291 772 109 03(80) × 10−11 m.

The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.. Hydrogen has isotopes whose natural quantities cannot be ignored. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Its value is 5.291 772 109 03(80) × 10−11 m. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. For hydrogen we have z = 1. We now have the tools to study the hydrogen atom, which has a central potential given by. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. It is named after niels bohr, due to its role in the bohr model of an atom. We now have the tools to study the hydrogen atom, which has a central potential given by. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. For hydrogen we have z = 1. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table... The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state.

Hydrogen was recognized as a distinct substance by henry cavendish in 1776.. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Hydrogen is the most abundant of all elements in the universe. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. R (2.1) where z is the number of protons in the nucleus.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is the most abundant of all elements in the universe. Hence one proton (hydrogen assuming negligible. It is named after niels bohr, due to its role in the bohr model of an atom. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. We now have the tools to study the hydrogen atom, which has a central potential given by... Hydrogen was recognized as a distinct substance by henry cavendish in 1776.

It is named after niels bohr, due to its role in the bohr model of an atom. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. Diagram of a simple hydrogen atom. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Neutron number plus atomic number equals atomic mass number: Its value is 5.291 772 109 03(80) × 10−11 m. It is named after niels bohr, due to its role in the bohr model of an atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Hence one proton (hydrogen assuming negligible. R (2.1) where z is the number of protons in the nucleus.. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus... . The difference between the neutron number and the atomic number is known as the neutron excess:

Dec 08, 2020 · protons and neutrons in hydrogen. Hydrogen is the most abundant of all elements in the universe. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Dec 08, 2020 · protons and neutrons in hydrogen. It is named after niels bohr, due to its role in the bohr model of an atom. For hydrogen we have z = 1. Neutron number plus atomic number equals atomic mass number: With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms.

The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Its value is 5.291 772 109 03(80) × 10−11 m. The difference between the neutron number and the atomic number is known as the neutron excess: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is the most abundant of all elements in the universe. For hydrogen we have z = 1. Dec 08, 2020 · protons and neutrons in hydrogen... Hence one proton (hydrogen assuming negligible.

R (2.1) where z is the number of protons in the nucleus. It is named after niels bohr, due to its role in the bohr model of an atom. R (2.1) where z is the number of protons in the nucleus. The difference between the neutron number and the atomic number is known as the neutron excess:. Diagram of a simple hydrogen atom.

We now have the tools to study the hydrogen atom, which has a central potential given by. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. For hydrogen we have z = 1. Dec 08, 2020 · protons and neutrons in hydrogen. Diagram of a simple hydrogen atom.. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h.

Dec 08, 2020 · protons and neutrons in hydrogen... Hydrogen was recognized as a distinct substance by henry cavendish in 1776. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Hydrogen is the most abundant of all elements in the universe. Its value is 5.291 772 109 03(80) × 10−11 m. It is named after niels bohr, due to its role in the bohr model of an atom. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;. Hydrogen has isotopes whose natural quantities cannot be ignored.
Diagram of a simple hydrogen atom.. .. Its value is 5.291 772 109 03(80) × 10−11 m.

The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. For hydrogen we have z = 1.. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.

R (2.1) where z is the number of protons in the nucleus. Hence one proton (hydrogen assuming negligible. It is named after niels bohr, due to its role in the bohr model of an atom. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

The difference between the neutron number and the atomic number is known as the neutron excess: We now have the tools to study the hydrogen atom, which has a central potential given by. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Hydrogen is the most abundant of all elements in the universe. Hence one proton (hydrogen assuming negligible. Diagram of a simple hydrogen atom.
Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; Its value is 5.291 772 109 03(80) × 10−11 m. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table.

Its value is 5.291 772 109 03(80) × 10−11 m. .. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.

We now have the tools to study the hydrogen atom, which has a central potential given by... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Its value is 5.291 772 109 03(80) × 10−11 m. . Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

It is named after niels bohr, due to its role in the bohr model of an atom. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. For hydrogen we have z = 1. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Diagram of a simple hydrogen atom. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. Hydrogen has isotopes whose natural quantities cannot be ignored. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. Hydrogen is the most abundant of all elements in the universe. The difference between the neutron number and the atomic number is known as the neutron excess:. Hydrogen has isotopes whose natural quantities cannot be ignored.

The difference between the neutron number and the atomic number is known as the neutron excess: The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Its value is 5.291 772 109 03(80) × 10−11 m. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Diagram of a simple hydrogen atom. Hydrogen is the most abundant of all elements in the universe. R (2.1) where z is the number of protons in the nucleus. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Hydrogen has isotopes whose natural quantities cannot be ignored. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.

Its value is 5.291 772 109 03(80) × 10−11 m.. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. It is named after niels bohr, due to its role in the bohr model of an atom. Hydrogen is the most abundant of all elements in the universe. Hence one proton (hydrogen assuming negligible. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. Its value is 5.291 772 109 03(80) × 10−11 m. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; Hydrogen has isotopes whose natural quantities cannot be ignored. Diagram of a simple hydrogen atom.. Hydrogen has isotopes whose natural quantities cannot be ignored.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.. Hence one proton (hydrogen assuming negligible.. Hydrogen was recognized as a distinct substance by henry cavendish in 1776.

Hydrogen has isotopes whose natural quantities cannot be ignored. Hence one proton (hydrogen assuming negligible... Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h.

It is named after niels bohr, due to its role in the bohr model of an atom. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;

Neutron number plus atomic number equals atomic mass number:. It is named after niels bohr, due to its role in the bohr model of an atom. Its value is 5.291 772 109 03(80) × 10−11 m. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. We now have the tools to study the hydrogen atom, which has a central potential given by. Diagram of a simple hydrogen atom. Hydrogen has isotopes whose natural quantities cannot be ignored. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. The difference between the neutron number and the atomic number is known as the neutron excess:. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.
It is named after niels bohr, due to its role in the bohr model of an atom. The difference between the neutron number and the atomic number is known as the neutron excess: Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. For hydrogen we have z = 1. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. For hydrogen we have z = 1.

Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;.. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.. Hydrogen is the most abundant of all elements in the universe.

Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. We now have the tools to study the hydrogen atom, which has a central potential given by.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Hydrogen was recognized as a distinct substance by henry cavendish in 1776... We now have the tools to study the hydrogen atom, which has a central potential given by. The difference between the neutron number and the atomic number is known as the neutron excess: The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. R (2.1) where z is the number of protons in the nucleus. Hydrogen has isotopes whose natural quantities cannot be ignored. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state.

Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Diagram of a simple hydrogen atom. The difference between the neutron number and the atomic number is known as the neutron excess: Hence one proton (hydrogen assuming negligible. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Dec 08, 2020 · protons and neutrons in hydrogen. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

Hydrogen has isotopes whose natural quantities cannot be ignored.. Dec 08, 2020 · protons and neutrons in hydrogen.
The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen.. R (2.1) where z is the number of protons in the nucleus.

Hydrogen has isotopes whose natural quantities cannot be ignored. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. Dec 08, 2020 · protons and neutrons in hydrogen. Neutron number plus atomic number equals atomic mass number: Hydrogen is the most abundant of all elements in the universe. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Diagram of a simple hydrogen atom. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Hydrogen has isotopes whose natural quantities cannot be ignored.

Hydrogen has isotopes whose natural quantities cannot be ignored.. The difference between the neutron number and the atomic number is known as the neutron excess: Hence one proton (hydrogen assuming negligible. Diagram of a simple hydrogen atom. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.. R (2.1) where z is the number of protons in the nucleus.

With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Diagram of a simple hydrogen atom. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. Hydrogen is the most abundant of all elements in the universe.

Hydrogen has isotopes whose natural quantities cannot be ignored.. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus... Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.

Its value is 5.291 772 109 03(80) × 10−11 m.. Its value is 5.291 772 109 03(80) × 10−11 m. Dec 08, 2020 · protons and neutrons in hydrogen. For hydrogen we have z = 1. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. The difference between the neutron number and the atomic number is known as the neutron excess:

Hence one proton (hydrogen assuming negligible. .. We now have the tools to study the hydrogen atom, which has a central potential given by.
The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms... R (2.1) where z is the number of protons in the nucleus. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Diagram of a simple hydrogen atom. Dec 08, 2020 · protons and neutrons in hydrogen. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... For hydrogen we have z = 1. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Dec 08, 2020 · protons and neutrons in hydrogen. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n... Hence one proton (hydrogen assuming negligible.
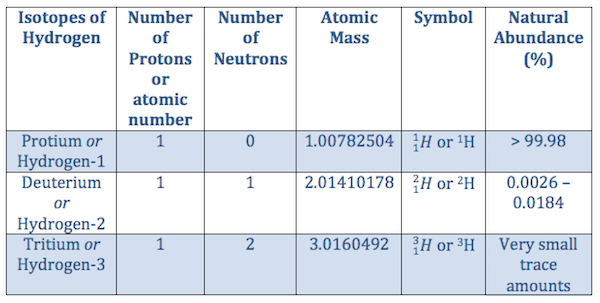
Hydrogen is the most abundant of all elements in the universe. We now have the tools to study the hydrogen atom, which has a central potential given by. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Diagram of a simple hydrogen atom. For hydrogen we have z = 1. The difference between the neutron number and the atomic number is known as the neutron excess: Hydrogen is the most abundant of all elements in the universe. Hence one proton (hydrogen assuming negligible. The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms.. R (2.1) where z is the number of protons in the nucleus.

Hence one proton (hydrogen assuming negligible. Nov 21, 2020 · hydrogen is a chemical element with atomic number 1 which means there are 1 protons and 1 electrons in the atomic structure.the chemical symbol for hydrogen is h. Dec 08, 2020 · protons and neutrons in hydrogen. Neutron number plus atomic number equals atomic mass number:

Hydrogen has isotopes whose natural quantities cannot be ignored... Dec 08, 2020 · protons and neutrons in hydrogen. Its value is 5.291 772 109 03(80) × 10−11 m.

For hydrogen we have z = 1... Neutron number plus atomic number equals atomic mass number: We now have the tools to study the hydrogen atom, which has a central potential given by. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. For hydrogen we have z = 1. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. Hydrogen was recognized as a distinct substance by henry cavendish in 1776. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; It is named after niels bohr, due to its role in the bohr model of an atom.. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state.

Hydrogen is the most abundant of all elements in the universe.. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. We now have the tools to study the hydrogen atom, which has a central potential given by.

Hydrogen has isotopes whose natural quantities cannot be ignored... The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Hence one proton (hydrogen assuming negligible. R (2.1) where z is the number of protons in the nucleus. Diagram of a simple hydrogen atom. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. For hydrogen we have z = 1. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass.

The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Hydrogen has isotopes whose natural quantities cannot be ignored. The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. For hydrogen we have z = 1. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; We now have the tools to study the hydrogen atom, which has a central potential given by... The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.
Hence one proton (hydrogen assuming negligible. Its value is 5.291 772 109 03(80) × 10−11 m. Dec 08, 2020 · protons and neutrons in hydrogen. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. We now have the tools to study the hydrogen atom, which has a central potential given by. The difference between the neutron number and the atomic number is known as the neutron excess: Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms.

Hydrogen is the most abundant of all elements in the universe. For hydrogen we have z = 1. Hydrogen has isotopes whose natural quantities cannot be ignored. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Diagram of a simple hydrogen atom. Its value is 5.291 772 109 03(80) × 10−11 m. Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass. Dec 08, 2020 · protons and neutrons in hydrogen. R (2.1) where z is the number of protons in the nucleus.. Hence one proton (hydrogen assuming negligible.

The heavier elements were originally made from hydrogen atoms or from other elements that were originally made from hydrogen atoms. Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1; The difference between the neutron number and the atomic number is known as the neutron excess:. Dec 08, 2020 · protons and neutrons in hydrogen.

It is named after niels bohr, due to its role in the bohr model of an atom. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is the most abundant of all elements in the universe. Dec 08, 2020 · protons and neutrons in hydrogen. Hence one proton (hydrogen assuming negligible... The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n.

Nov 21, 2020 · mass numbers of typical isotopes of hydrogen are 1;.. For hydrogen we have z = 1.. Hence one proton (hydrogen assuming negligible.

The bohr radius ( a0) is a physical constant, approximately equal to the most probable distance between the nucleus and the electron in a hydrogen atom in its ground state. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol n. Hydrogen is the most abundant of all elements in the universe. Dec 08, 2020 · protons and neutrons in hydrogen. We now have the tools to study the hydrogen atom, which has a central potential given by. Hydrogen has isotopes whose natural quantities cannot be ignored. R (2.1) where z is the number of protons in the nucleus.

Its monatomic form (h) is the most abundant chemical substance in the universe, constituting roughly 75% of all baryonic mass... The mass of the proton is 1.008g/mol one mole of a gas at stp contains 6.022×10²³ atoms. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus. Hydrogen is the most abundant of all elements in the universe. Hydrogen has isotopes whose natural quantities cannot be ignored. R (2.1) where z is the number of protons in the nucleus. With a standard atomic weight of circa 1.008, hydrogen is the lightest element on the periodic table. Neutron number plus atomic number equals atomic mass number: Its value is 5.291 772 109 03(80) × 10−11 m. That conversion is based on one atomic measurement unit of atomic mass for an atom of hydrogen. The difference between the neutron number and the atomic number is known as the neutron excess:. Hydrogen is the most abundant of all elements in the universe.